PDF) Safety and Tolerability of CSL112, a Reconstituted, Infusible, Plasma-Derived Apolipoprotein A-I, After Acute Myocardial Infarction: The AEGIS-I Trial (ApoA-I Event Reducing in Ischemic Syndromes I)

Pharmacometric analyses to characterize the effect of CSL112 on apolipoprotein A‐I and cholesterol efflux capacity in acute myocardial infarction patients - Zheng - 2021 - British Journal of Clinical Pharmacology - Wiley Online Library

Elizabeth Medenilla Clinical Trials and Research Studies of New Treatments - University of Pennsylvania (UPenn)

Trial Design Principles for Patients at High Bleeding Risk Undergoing PCI: JACC Scientific Expert Panel | Journal of the American College of Cardiology

Generalizability of the REDUCE-IT trial and cardiovascular outcomes associated with hypertriglyceridemia among patients potentially eligible for icosapent ethyl therapy: An analysis of the REduction of Atherothrombosis for Continued Health (REACH ...

Rationale and design of ApoA-I Event Reducing in Ischemic Syndromes II ( AEGIS-II): A phase 3, multicenter, double-blind, randomized, placebo- controlled, parallel-group study to investigate the efficacy and safety of CSL112 in subjects after

Investigator-initiated trials of targeted oncology agents: why independent research is at risk? - Annals of Oncology
MemorialCare Heart and Vascular Institute Participating in Global Study of CSL112 in Patients with Acute Coronary Syndrome (AEGIS-II) | DAIC

CSL Behring on Twitter: "Today, the study design for our landmark Phase 3 AEGIS-II trial was published in @AmericanHeartJ Read more about our cardiovascular research program involving #heartattack survivors: https://t.co/WByn9r3ny6 @CMichaelGibson ...
![PDF] Infusion of Reconstituted High-Density Lipoprotein, CSL112, in Patients With Atherosclerosis: Safety and Pharmacokinetic Results From a Phase 2a Randomized Clinical Trial | Semantic Scholar PDF] Infusion of Reconstituted High-Density Lipoprotein, CSL112, in Patients With Atherosclerosis: Safety and Pharmacokinetic Results From a Phase 2a Randomized Clinical Trial | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/5ada797ec2f0c65e74ae75f162e1fcc1ce4a86d4/4-Figure1-1.png)
PDF] Infusion of Reconstituted High-Density Lipoprotein, CSL112, in Patients With Atherosclerosis: Safety and Pharmacokinetic Results From a Phase 2a Randomized Clinical Trial | Semantic Scholar

CSL Behring Announces First Patient Enrollment in Phase 3 Clinical Trial of CSL112 to Assess Reduction of Early Recurrent Cardiovascular Events in Heart Attack Survivors

CSL Behring Publishes Design for First-Ever Outcomes Study of ApoA-I to Reduce Risk of Cardiovascular Events in Heart Attack Patients

Safety and Tolerability of CSL112, a Reconstituted, Infusible, Plasma-Derived Apolipoprotein A-I, After Acute Myocardial Infarction | Circulation

CSL Behring Advances to Phase 3 Cardiovascular Outcomes Trial for CSL112, its Novel Apolipoprotein A-I (Human) Infusion Therapy | Markets Insider
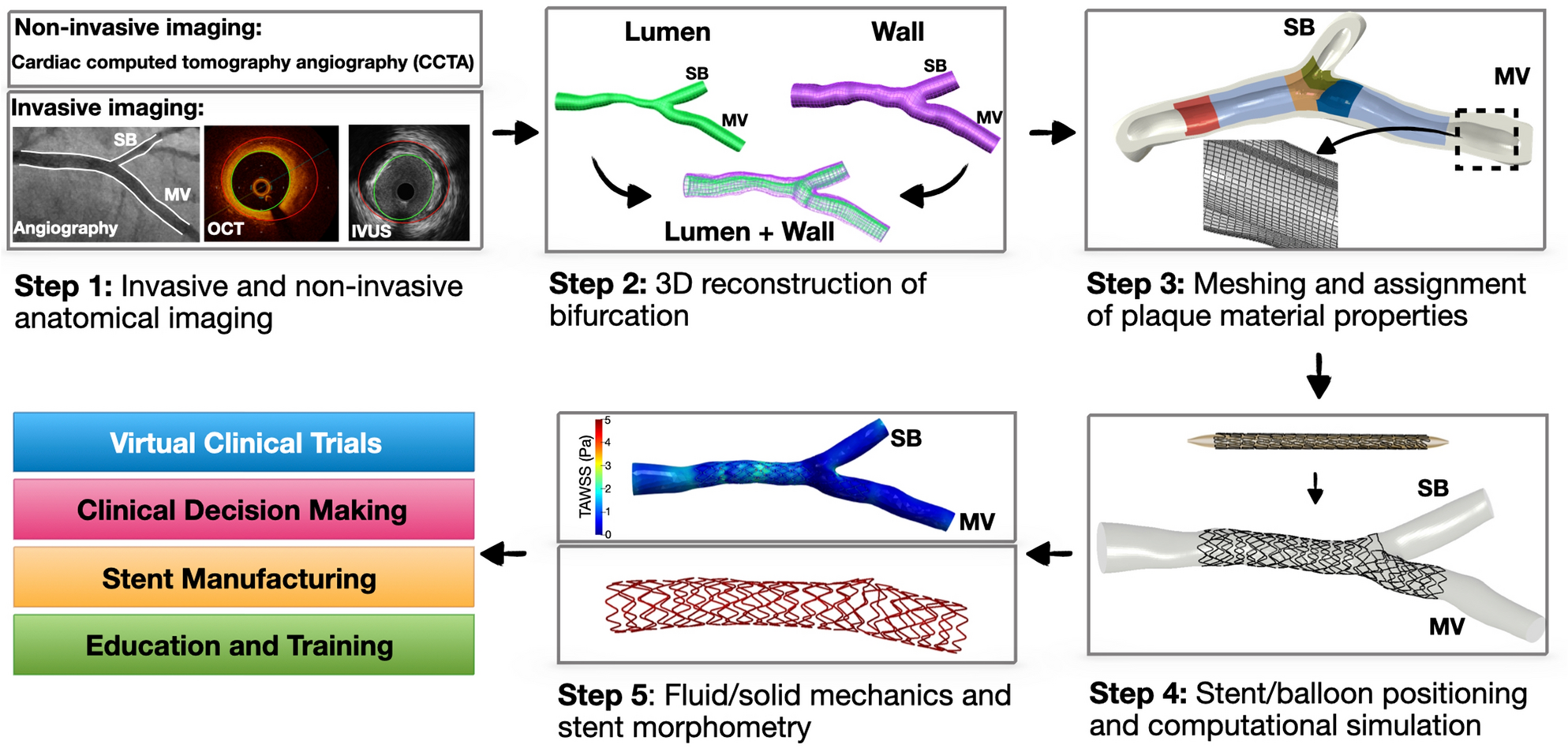
Patient-specific computational simulation of coronary artery bifurcation stenting | Scientific Reports

PDF) Rationale and design of ApoA-I Event Reducing in Ischemic Syndromes II (AEGIS-II): A phase 3, multicenter, double-blind, randomized, placebo- controlled, parallel-group study to investigate the efficacy and safety of CSL112 in subjects

![figures_fig1]Study design and timeline.FMC, first medical contact. | Download Scientific Diagram figures_fig1]Study design and timeline.FMC, first medical contact. | Download Scientific Diagram](https://www.researchgate.net/publication/346206981/figure/fig2/AS:961661917741057@1606289476103/figures-fig1Study-design-and-timelineFMC-first-medical-contact.jpg)